GATA/Heme Multi-omics Reveals a Trace Metal-Dependent Cellular Differentiation Mechanism.
GATA/Heme Multi-omics Reveals a Trace Metal-Dependent Cellular Differentiation Mechanism.
Abstract
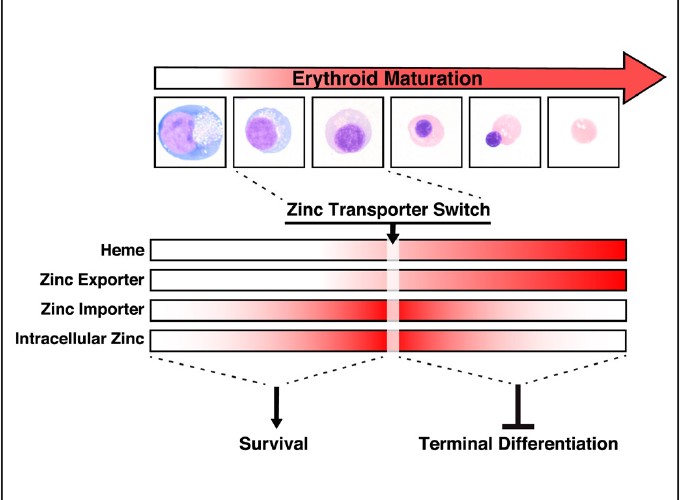
By functioning as an enzyme cofactor, hemoglobin component, and gene regulator, heme is vital for life. One mode of heme-regulated transcription involves amplifying the activity of GATA-1, a key determinant of erythrocyte differentiation. To discover biological consequences of the metal cofactor-transcription factor mechanism, we merged GATA-1/heme-regulated sectors of the proteome and transcriptome. This multi-omic analysis revealed a GATA-1/heme circuit involving hemoglobin subunits, ubiquitination components, and proteins not implicated in erythrocyte biology, including the zinc exporter Slc30a1. Though GATA-1 induced expression of Slc30a1 and the zinc importer Slc39a8, Slc39a8 dominantly increased intracellular zinc, which conferred erythroblast survival. Subsequently, a zinc transporter switch, involving decreased importer and sustained exporter expression, reduced intracellular zinc during terminal differentiation. Downregulating Slc30a1 increased intracellular zinc and, strikingly, accelerated differentiation. This analysis established a conserved paradigm in which a GATA-1/heme circuit controls trace metal transport machinery and trace metal levels as a mechanism governing cellular differentiation.